Essential Synopsis
An analysis of photophysical and photochemical effects in mega-networks of tryptophan in biological architectures has found collective coherence resulting in quantum optical enhancement, known as superradiance, and long-range interactivity even in the ambient thermal environment of the cell [1]. Such collective quantum optical effects in tryptophan mega-networks may underlie key cellular orchestration and signaling processes like potentially ultrafast information transfer in neuronal axons, which have highly ordered core microtubule filamentary bundles enriched with coordinated tryptophan moieties that are involved in information processing. Such empirical evidence supports theories like The Unified Spacememory Network [2] and the Orchestrated Objective Reduction (Orch-OR model) of Hameroff and Penrose [3], both of which involve unified physics concepts of spacetime micro-structure coupled with quantum states like those involving collective coherence of subcellular macromolecular assemblies such as microtubules and mitochondria (filamentary mitochondria couple with microtubules to form a mitochondrial reticular matrix), which underlie informational events correlated with cognitive processes and potentially awareness.
What was Found
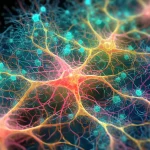
An international research team (from the USA, Mexico, Switzerland, and Italy) have performed a comprehensive analysis combining computational modeling, theoretical analysis, and experimental measurements to document and characterize collective optical signal processing in tryptophan mega-networks such as those found in microtubules. For example, a microtubule super-architecture, called a centriole (Figure 1), of 320 nm length will contain around 105 tryptophan chromophores (a chromophore is a molecule that absorbs and emits light). At such size, long-range resonance and synchronization of dipole oscillations makes protein architectures like this mega-networks and helps to explain previous studies like that by Bandopahdyay et al. [4] that have found significant coordination among single microtubules behaving as if the entire supramolecular assembly was in a quantum state, like a Frohlich condensate [5-8]. This latest study as well as the multiple studies by the Bandopahdyay research group are direct empirical validation of Hameroff and Penrose’s Orch-OR model of collective coherent quantum states underlying brain cognition [S. Hameroff and R. Penrose, “Orchestrated reduction of quantum coherence in brain microtubules: A model for consciousness,” Mathematics and Computers in Simulation, vol. 40, no. 3, pp. 453–480, Apr. 1996, doi: 10.1016/0378-4754(96)80476-9].
Therefore, such a finding supports theories of microtubules occupying essential roles in cellular information processing: possibly playing significant roles in cognitive processes like those involving neurons. The collective optical response of tryptophan networks in protein architectures is characterized by the absorption and emission spectra of the chromophores in the network, and analysis of this collective optical response of tryptophan networks has identified superradiant enhancements, a quantum effect that results in a higher emission intensity of coherent electromagnetic flux, which could be utilized by the cell for signaling and high efficiency energy transport.
The collective optical response can be affected by structural disorder, but the larger networks of coordinated tryptophan polymer residues examined in this study tend to exhibit cooperative robustness to disorder. This phenomenon involves a collection of identical tryptophan chromophores spontaneously emitting intense coherent radiation. The transition dipoles of the tryptophan chromophores synchronize coherently, leading to superradiant states that enhance excitation transfer within the network. This cooperative quantum effect results in a higher peak intensity of emission compared to that of a single chromophore. Thus, the collective photoexcitonic properties of large tryptophan networks in protein structures ranging from individual tubulin dimers and microtubule segments to microtubule super-architectures such as the centriole and neuronal bundles is evident in the measured fluorescent yield, which increases significantly from single tubulins to tubulin assemblies, a key feature of superradiance and thus collective quantum coherent properties.
The collective optical response is influenced by the coupling between the tryptophan chromophores induced by the electromagnetic field. The collective optical response of tryptophan networks in protein architectures is characterized by the quantum yield (QY) enhancements. The QY is a figure of merit for cooperativity and is a robust measure of the enhancement of the optical response. The QY is not significantly affected by static disorder, even for disorder strengths of the same magnitude as room-temperature energy. This robustness is attributed to the distribution of the superradiant dipole strength among other excitonic states, while states close to the superradiant state in energy still exhibit most of the dipole strength.
Therefore, the collective optical response of tryptophan networks in protein architectures is characterized by a persistent enhancement of the quantum yield, even under ambient conditions. The collective optical response of tryptophan networks in protein architectures is the formation of strongly superradiant states due to collective interactions among organized arrangements of up to more than 105 tryptophan UV-excited transition dipoles in microtubule architectures. This leads to an enhancement of the fluorescence quantum yield, which has been confirmed by experiments. The superradiant behavior in the fluorescence quantum yield is observed in hierarchically organized tubulin structures, such as microtubules, centrioles, and neuronal bundles. The quantum yield increases in different geometric regimes at thermal equilibrium before saturation, highlighting the persistence of the effect in the presence of disorder, such that the collective quantum coherent state is robust even at temperatures typical for the living cell.
Dr. Kurian and the Quantum Biology Lab
The study was led by Philip Kurian, the principle investigator and founder of the quantum biology lab at Howard University, where researchers explore fundamental questions at the nexus of quantum theory, electrodynamics, and biosystems. Kurian has previously performed collaborative studies with Hameroff, Craddock, and Tuszynski—all foremost leaders in the science of quantum biology—investigating questions like the quantum brain hypothesis (Figure 2).
In their paper Quantum Processes in Neurophotonics and the Origin of the Brain’s Spatiotemporal Hierarchy (T. J. A. Craddock, P. Kurian, J. A. Tuszynski, and S. R. Hameroff, 2019), the Hameroff research team argues that the brain’s complex nonlinear dynamics may allow quantum effects to influence neural activity across multiple scales. They discuss evidence for photon generation and transmission in neurons, potentially serving as quantum information carriers. The paper examines how reactive oxygen species in mitochondria may produce ultraweak photon emissions that interact with aromatic amino acid networks in proteins like tubulin. This could enable coherent energy transfer along microtubules, similar to quantum effects seen in photosynthesis. The authors propose this may form a basis for rapid intracellular signaling and contribute to cognitive processes.
Classical neural signaling pathways rely on the well-established principles of electrochemical transmission. Neurons communicate through synaptic transmission, where neurotransmitters are released from the presynaptic neuron and bind to receptors on the postsynaptic neuron. This process involves the propagation of action potentials along axons, driven by the movement of ions across cell membranes. The speed and fidelity of this communication are influenced by factors such as myelination and synaptic efficiency.
In contrast, neurophotonic mechanisms suggest that neurons might also use photons—particles of light—as a mode of information transfer. Unlike classical signals that depend on ion fluxes and chemical gradients, photonic communication could leverage quantum coherence and entanglement. This would allow for ultra-fast, non-local interactions that defy classical limitations. Photons generated within neurons could traverse intracellular distances with minimal loss of information, potentially synchronizing activities across different regions with unprecedented precision.
While classical pathways are constrained by the speed at which ions can move and synapses can release neurotransmitters, neurophotonic processes might operate at the speed of light, providing a much faster alternative route for neural communication. Additionally, photonic signaling could complement traditional mechanisms by integrating multiple layers of information processing—combining electrical, chemical, and photonic signals to create a more robust and versatile neural network.
The coexistence of these two modes might explain some aspects of brain function that remain puzzling under purely classical models. For example, how the brain achieves rapid integration of sensory inputs or maintains synchronous oscillations across distant regions during cognitive tasks could be better understood through this dual framework. Neurophotonic mechanisms offer an exciting frontier in neuroscience, promising to deepen our comprehension of both fundamental brain operations and higher-order mental functions.
How the Study was Performed
Experimental testing was performed using a combination of computational modeling, theoretical analysis, and experimental measurements. For computational modeling: protein structural models were created using atomic coordinates from a resource called the Protein Data Bank, from which transition dipole moment coordinates of tryptophan chromophores were extracted from the models. For the theoretical analysis: quantum optics and effective Hamiltonian approaches (in quantum mechanics, the Hamiltonian of a system is used to calculate the total energy of that system) were used to describe the interaction between tryptophan networks and the electromagnetic field. Theoretical models were developed based on dipole-dipole correlation functions and the Boltzmann factor (used to calculate things like the energy difference between two magnetic energy levels), from which complex eigenvalues of the radiative Hamiltonian were solved to determine radiative decay rates and superradiant enhancement factors. Predictions were then made for the collective optical response and fluorescence quantum yields of tryptophan networks.
With computer and theoretical modeling at hand, the researchers performed the all-important experimental measurements to test key predictions based on the calculations. Steady-state spectroscopy was performed on tryptophan networks in protein architectures and absorption and fluorescence spectra were measured to obtain parameters such as absorption maxima, fluorescence maxima, and quantum yields. The measured quantum yields were compared with theoretical predictions to validate the collective optical response.
The theoretical predictions were compared with experimental data to assess the accuracy of the models and the effects of structural disorder on the collective optical response were investigated. As well the robustness of quantum yield enhancements to static disorder was analyzed. After comprehensive analysis the research team were able to use their data to validate the predictions based on the theoretical modeling and demonstrated a positive effect of collective quantum behavior resulting in non-trivial superradiance of tryptophan chromophores in macromolecules like microtubules.
The Significance of Tryptophan in the Biological System
Tryptophan plays a multifaceted role in biological signaling [9]. It is involved in the synthesis of proteins and serves as a key component of signaling molecules and neurotransmitters such as serotonin. Tryptophan also acts as a precursor for niacin and is involved in energy metabolism and DNA repair. It can act as a chromophore, participating in light absorption and emission processes, and is used as a probe for studying protein-protein interactions and conformational changes. Tryptophan is also involved in the regulation of gene expression and can be altered in certain diseases and disorders. Overall, tryptophan’s unique properties make it a versatile molecule with diverse functions in various physiological processes.
Trp mega-networks support quantum states in proteins through collective and cooperative effects. When multiple Trp chromophores are organized in a network (Figure 3), their transition dipoles can interact with each other, leading to the emergence of superradiant and subradiant eigenmodes. These eigenmodes represent collective excitations of the Trp chromophores, where the excitation energy is shared and distributed among the network.
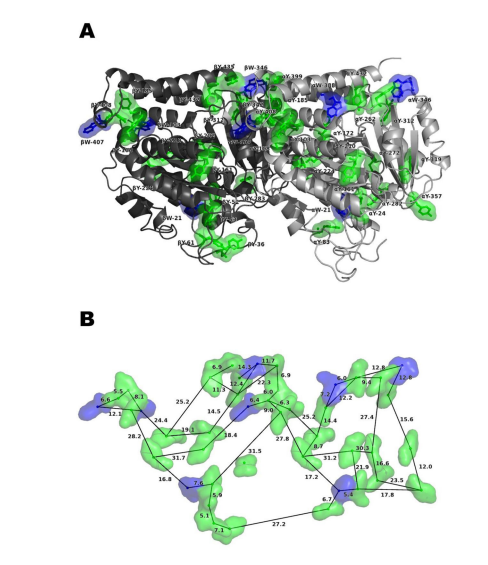
The coupling between Trp transition dipoles in the mega-networks allows for long-range interactions, even though the individual couplings are relatively weak compared to room-temperature energy. This long-range coupling enhances the robustness of the network and protects it from disorder. The strong coupling between the Trp chromophores and the electromagnetic field promotes the formation of superradiant states, where the emission rate is significantly enhanced compared to that of individual Trp chromophores.
The superradiant enhancement factor, which compares the radiative decay rate of the brightest state in the network to that of a single Trp chromophore, increases with the size of the network until it reaches a saturation point. This means that larger Trp mega-networks can exhibit stronger superradiant effects. The presence of these superradiant states in various biological complexes, such as microtubules and centrioles, is confirmed by theoretical analysis and experimental measurements of fluorescence quantum yield.
These collective quantum optical effects in Trp mega-networks have important implications for biological systems. They can enhance the absorption and energy transfer of UV photoexcitations, promote photoprotection by re-emitting absorbed energy, and potentially facilitate ultrafast information transfer in neuronal axons. The robustness of these quantum states to thermal equilibrium and structural disorder further highlights their potential significance in cellular signaling and control. Note that molecules enriched in tryptophan moieties include cytochrome c of the mitochondria, so that this organelle is likely to play a similar and important role in the quantum optical signaling system of the cell.
Key Findings of the Study:
- Collective Quantum States:
- Trp chromophores, when organized into networks, interact through their transition dipoles. This interaction leads to the formation of collective excitations known as superradiant and subradiant eigenmodes.
- In superradiant states, excitation energy is distributed across the network, leading to significantly enhanced emission rates compared to individual Trp molecules.
- Long-Range Coupling:
- Despite the relatively weak individual couplings between Trp transition dipoles, the overall network exhibits long-range interactions. This enhances the robustness of the network against structural disorder and thermal fluctuations.
- Superradiant Enhancement Factor:
- The radiative decay rate of the brightest state in the network is much higher than that of a single Trp chromophore. This superradiant enhancement increases with the size of the network until a saturation point is reached.
- Presence in Biological Structures:
- Superradiant states are found in various biological complexes, such as microtubules and centrioles. These findings are supported by theoretical analysis and experimental measurements of fluorescence quantum yield.
Implications for Biological Systems:
- Enhanced UV Absorption and Energy Transfer:
- The superradiant properties of Trp networks enhance the absorption and transfer of UV photoexcitations. This could improve the efficiency of energy utilization in biological systems.
- Photoprotection:
- The networks can re-emit absorbed energy, potentially protecting cells from UV-induced damage by dissipating excess energy efficiently.
- Ultrafast Information Transfer:
- The robustness and efficiency of these quantum states suggest a potential role in facilitating rapid information transfer within neuronal axons and other cellular structures.
- Thermal and Structural Robustness:
- The quantum states in Trp mega-networks are resilient to thermal fluctuations and structural disorder, highlighting their importance in maintaining cellular signaling and control mechanisms under varying conditions.
The study demonstrates that tryptophan, beyond its well-known roles in protein synthesis, neurotransmission, and metabolic pathways, also participates in sophisticated quantum optical phenomena when organized into mega-networks. These networks’ superradiant properties could have significant implications for enhancing energy transfer, photoprotection, and information processing in biological systems. The findings open new avenues for understanding the quantum aspects of biological processes and their potential applications in biotechnology and medicine.
Potential Insights to Glean from the Study
From the theoretical modeling and experimental validation, the study demonstrates the potential of significant photophysical and photochemical effects in mega-networks of tryptophan in biological architectures resulting from quantum optical enhancement during collective coherence, long-range resonance and synchronization. Contrary to what used to be the common sentiment among experts that the biological system was too wet, noisy, and warm for non-trivial quantum effects like collective coherence, the observed superradiance and long-range interactivity even in the ambient thermal environment of the cell disprove such naïve perspectives (naïve because they were not taking into full account the unique structural / geometric organizational properties of cellular proteins and were based on very simplistic purviews of cellular organization).
The findings have wide-ranging implications and are important because collective quantum optical effects in tryptophan mega-networks may underlie key cellular orchestration and signaling processes, like potentially ultrafast information transfer in neuronal axons, which have highly ordered core microtubule filamentary bundles (Figure 4). Indeed, the laser-like optical activity of microtubules, described as superradiance in the study by Kurian et al., has been observed in independent studies like that by the Bandyophadyay research group that describe how optical vortices originating within the core microtubule filamentary bundle of neurons transmit through an actin grid network below the neuronal membrane and directly regulate coherently vibrating ion channels to set precise timing of ion flux, resulting in microsecond regulation of millisecond nerve cell electrical conduction [10].
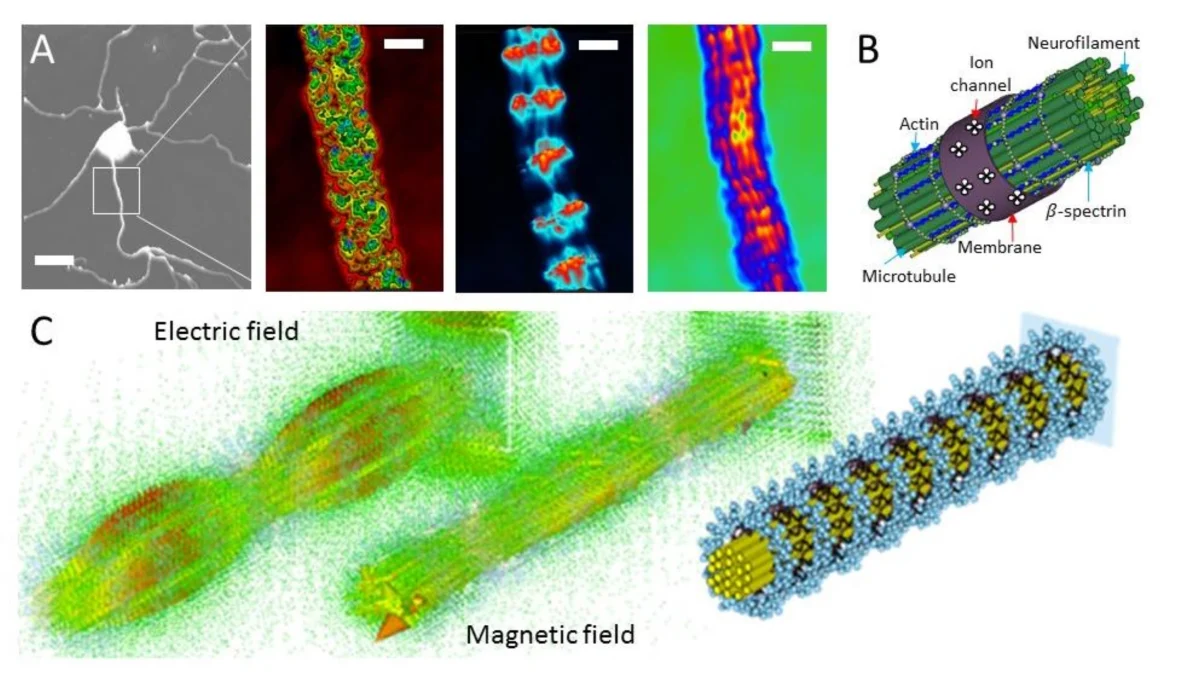
Such empirical evidence supports theories like The Unified Spacememory Network of Haramein and Brown, or Orchestrated Objective Reduction of Hameroff and Penrose, which involve subcellular macromolecular assemblies like mitochondria and microtubules (the mitochondrial reticular matrix). As an example, in regards to Hameroff-Penrose Orch-OR theory, popular science commentator physicist Sabina Hossenfelder recently commented: “When Roger Penrose originally came out with the idea that the human brain uses quantum effects in microtubules and that was the origin of consciousness, many thought the idea was a little crazy. According to a new study, it turns out that Penrose was actually right… about the microtubules anyways. Let’s have a look.”
In our work at the ISF on Spacememory Network Theory, we evaluated quantum collective coherence and the entanglement of dipoles of the aromatic amino acids like tryptophan in microtubules within the Unified Physics framework of equivalence between quantum entanglement and multiply connected spacetime geometries, as described in the Maldacena-Susskind holographic correspondence conjecture that Einstein-Rosen bridges are Einstein-Rosen-Podolsky correlations (ER = EPR). From this equivalency, we postulated that the collective quantum coherent and entanglement states of electronic dipoles in tubulin residues function in memory and this memory property is the coupling of quantum entanglement with the multiply connected geometry of space that results in the more general property of Spacememory.
In Spacememory Network Theory the Penrose-Lucas argument is supported because it is described how the property of memory, associated with consciousness, is not the result of computational processes of the brain but is a direct accession of information that corresponds with past spacetime coordinates in a organism’s worldline via quantum entanglement of dipole moments in tubulin residues. Wherein ER = EPR, or Einstein-Rosen bridges are quantum entanglement, and quantum entanglement links spacetime coordinates in an organism’s worldline via wormhole geometry. So, quantum entanglement links the informational states of microtubules not only with nonlocal spatial correlations but nonlocal temporal correlations as well.
From both examples, it is evident the importance of non-trivial quantum effects in the information, cognitive, and possibly awareness properties of the living system. This most recent study provides further validation of the previously theoretical supposition that non-trivial quantum mechanical states can be operational within the living system; an idea that was derided as “crazy” and “crackpot” for a long time by scientists who understood well the susceptibility of collective quantum states to decoherence but did not understand well the organization and energetic dynamics of the subcellular macromolecular systems of the living organism, which have evolved to preserve robustness of quantum states at physiological temperatures. Given the remarkable scale-free intelligence exhibited by the living system, it should be no surprise that life has leveraged non-trivial quantum mechanical phenomena for its own benefit, or even possibly emerged from such quantum properties as collective long-range resonance, coherence, and synchronization (see, for example the article Consciousness came before life).
References
[1] N. S. Babcock, G. Montes-Cabrera, K. E. Oberhofer, M. Chergui, G. L. Celardo, and P. Kurian, “Ultraviolet Superradiance from Mega-Networks of Tryptophan in Biological Architectures,” J. Phys. Chem. B, vol. 128, no. 17, pp. 4035–4046, May 2024, doi: 10.1021/acs.jpcb.3c07936.
[2] N. Haramein, W. D. Brown, and A. Val Baker, “The Unified Spacememory Network: from Cosmogenesis to Consciousness,” Neuroquantology, vol. 14, no. 4, Jun. 2016, doi: 10.14704/nq.2016.14.4.961.
[3] S. Hameroff and R. Penrose, “Orchestrated reduction of quantum coherence in brain microtubules: A model for consciousness,” Mathematics and Computers in Simulation, vol. 40, no. 3, pp. 453–480, Apr. 1996, doi: 10.1016/0378-4754(96)80476-9.
[4] S. Sahu et al., “Atomic water channel controlling remarkable properties of a single brain microtubule: correlating single protein to its supramolecular assembly,” Biosens Bioelectron, vol. 47, pp. 141–148, Sep. 2013, doi: 10.1016/j.bios.2013.02.050.
[5] Frohlich, H. Long-range coherence and energy storage in biological systems. Int. J. Quantum Chem. 1968, 2, 641–649.
[6] Frohlich, H. Quantum mechanical concepts in biology. In From Theoretical Physics to Biology; Marois, M., Ed.; North-Holland: Amsterdam, the Netherlands, 1969; pp. 13–22.
[7] Frohlich, H. Long range coherence and the action of enzymes. Nature 1970, doi:10.1038/2281093a0.
[8] Vasconcellos, Áurea & Vannucchi, Fabio & Oliveira, Sergio & Luzzi, Roberto. (2012). Fröhlich Condensate: Emergence of Synergetic Dissipative Structures in Information Processing Biological and Condensed Matter Systems. Information. 3. 601-620. 10.3390/info3040601.
[9] S. Barik, “The Uniqueness of Tryptophan in Biology: Properties, Metabolism, Interactions and Localization in Proteins,” Int J Mol Sci, vol. 21, no. 22, p. 8776, Nov. 2020, doi: 10.3390/ijms21228776.
[10] ] P. Singh et al., “Cytoskeletal Filaments Deep Inside a Neuron Are not Silent: They Regulate the Precise Timing of Nerve Spikes Using a Pair of Vortices,” Symmetry, vol. 13, no. 5, Art. no. 5, May 2021, doi: 10.3390/sym13050821.